LATIS® Spacer

Product Overview
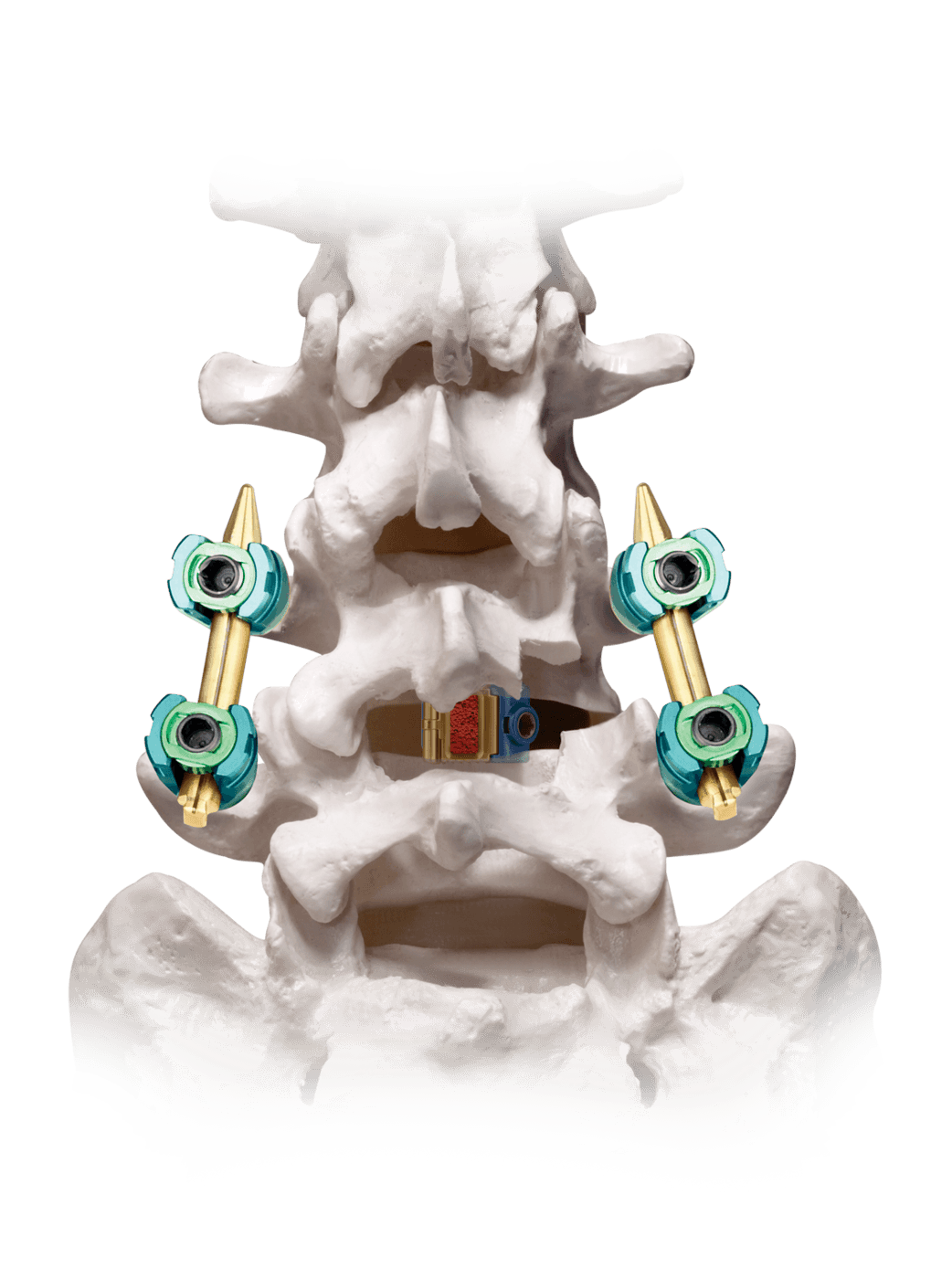
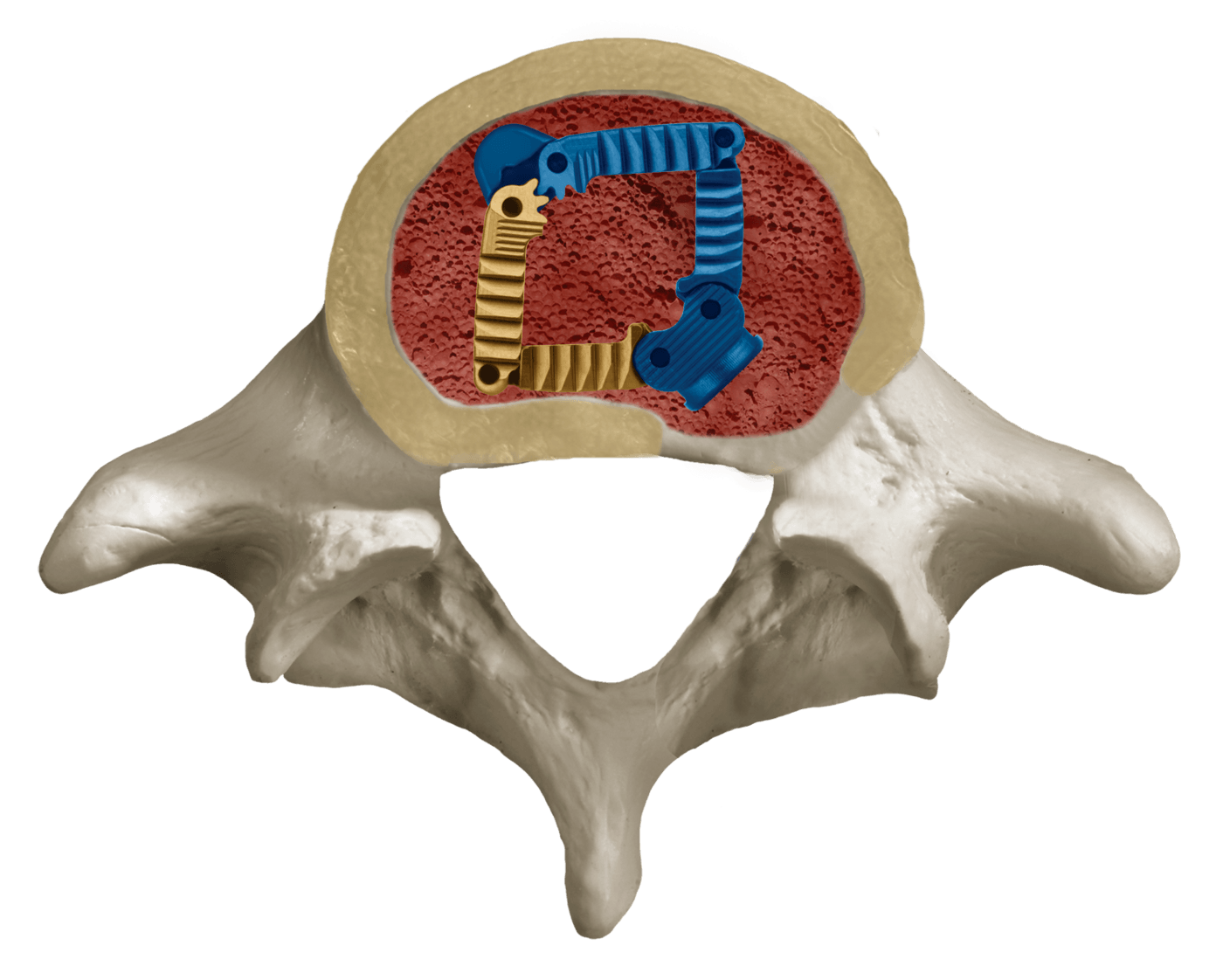
LATIS® is an innovative laterally expanding interbody spacer designed to provide the benefits of an ALIF spacer through a TLIF approach. This unique design maximizes implant footprint, surface area, and graft volume from a posterior approach.
Features and Benefits
MIS Approach, Maximal Footprint
Expands laterally in situ, optimizing apophyseal ring engagement to help reduce reduce subsidence.
Large Fusion Bed
Once expanded, a vast single graft chamber is created allowing for a distinctively large fusion mass.
Migration Resistance
The expanded implant geometry and slotted tooth pattern are designed to help resist implant migration.